Latest Articles
Replacements for 3M™ Novec™ Solvents from EnviroTech Europe

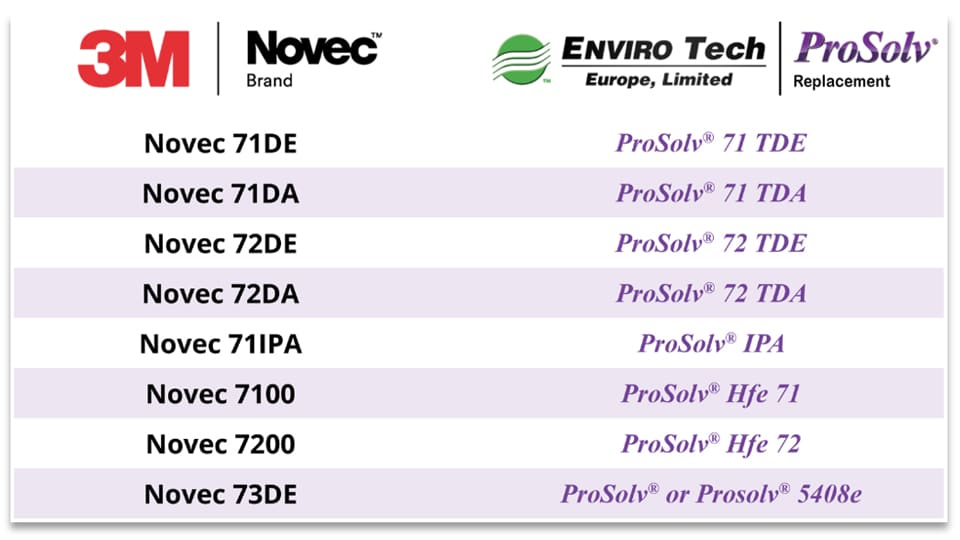
A range of drop-in replacements for 3M™ Novec™ solvents
In December 2022, 3M™ announced their plans to stop producing per- and polyfluoroalkyl substances (PFAS) in its product line by the end of 2025. This phase-out affects the following 3M™ product lines: 3M™ Novec™ Engineered Fluids 3M™ Fluorinert™ Electronic LiquidsARE YOU AFFECTED BY THE PHASE-OUT OF 3M™ NOVEC™ ENGINEERED FLUIDS?
At Envirotech Europe we can offer replacements with our ProSolv® range for all 3M™ Novec™ solvent cleaning fluids. We understand the uniqueness of every precision cleaning application and provide free on-site visits, technical support and cleaning trials when required.
We can provide a full assessment of your current precision cleaning process to put in a plan to help ensure a seamless transition.
Future-proof your cleaning process with our new ProSolv® range additions.
We have added several cost effective and efficient precision cleaning process formulations to our range of ProSolv® vapour degreasing solvents, offering an immediate drop in replacements for the 3M™ Novec™ engineered fluids.
Introducing ProSolv®5408e, a next generation PFAS free fluorinated solvent offering low GWP and zero ODP.
ProSolv® 5408e is a next generation PFAS free fluorinated solvent offering low Global Warming Potential (GWP) & zero Ozone Depletion Potential (ODP). A general multi-purpose vapour degreasing cleaning solvent that can act as an immediate drop-in replacement for most of the 3M™ Novec™ engineering fluids. We can also deliver an exact formulation replacement of your current solvent should that be your requirements.
EnviroTech Europe have many years of experience and our experts are available to guide you through your solvent cleaner changeover procedures or to advise on equipment. We have extensive partnerships with equipment manufacturers through our global distributors and can provide information and advice on solvents and solvent systems.
FURTHER INFORMATION
Please visit our website https://www.envirotech-europe.com/prosolv5408e for information about other uses and applications for ProSolv® 5408e.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
For more advice, please telephone us on +44 (0) 20 8281 6370 or use our website contact form. All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Related Posts
The importance of corrosion protection for electric vehicles

The importance of corrosion protection for electric vehicles
Corrosion protection is as important for electronic vehicles (EVs) as it is in traditional petrol/gasoline powered vehicles. However, there are some unique factors to consider when it comes to avoiding corrosion problems in electric vehicles:
Battery Enclosure and Connections:
EVs have large, heavy battery packs that are typically enclosed in a protective housing. This enclosure and its connections can be particularly susceptible to corrosion, especially in regions with high humidity or exposure to road salt. Corrosion in the battery enclosure or on electrical connections can affect the performance and safety of the vehicle.
Charging Infrastructure:
Charging equipment, especially in public charging stations, can be exposed to the elements. The connectors, plugs, and wiring can corrode over time. This can lead to issues with charging reliability and safety concerns.
Cooling Systems:
Many EVs use liquid cooling systems to manage the temperature of the battery. The components in these systems, such as radiators and coolant lines, are vulnerable to corrosion. The resulting leaks or blockages in the cooling system can potentially lead to overheating and reduced battery life.
Underbody and Frame:
Just like traditional vehicles, EVs have underbody components such as the chassis, suspension, and brake components that are susceptible to corrosion, especially if the vehicle is driven in areas where roads are salted during winter. Corrosion can weaken the structural integrity of the vehicle.
Electrical Contacts and Connectors:
Electrical connectors and contacts throughout the vehicle can corrode, leading to issues with sensors, lights, and other electrical systems. This can affect overall performance and safety.
Material Longevity:
EVs often incorporate a variety of materials, including metals and alloys, in their construction. Corrosion can significantly reduce the lifespan of these materials, leading to premature failure of crucial components. Protecting against corrosion ensures the durability and reliability of the vehicle over time.
Safety Concerns:
Corrosion can compromise the structural integrity of a vehicle, posing safety risks for occupants. In an EV, where advanced technologies and high-voltage systems are prevalent, maintaining structural integrity becomes even more critical. Proper corrosion protection helps mitigate these concerns.
Performance and Efficiency:
Corrosion on electrical components, connectors, and conductive elements can hinder the performance and efficiency of an EV. For instance, corrosion on battery terminals may increase electrical resistance, leading to reduced energy transfer efficiency and overall performance. Ensuring corrosion-free connections is vital for maintaining optimal efficiency.
Cost Savings:
Corrosion-related damage can result in expensive repairs and replacements. By investing in effective corrosion protection measures, manufacturers and owners can avoid these costs, contributing to the overall economic viability of electric vehicles.
Environmental Impact:
EVs are often considered more environmentally friendly than traditional vehicles. However, if corrosion leads to premature scrapping of EVs or frequent replacement of components, it can undermine their environmental benefits. Proper corrosion protection helps maintain the longevity of EVs, reducing their overall environmental impact.
Maintaining Resale Value:
Corrosion damage can significantly reduce the resale value of a vehicle. As EVs become more common in the used car market, maintaining corrosion protection becomes essential for preserving the value of these vehicles over time.
Regulatory Compliance:
Different regions have regulations and standards regarding vehicle safety. Corrosion protection is often a requirement to meet these standards and ensure that EVs adhere to guidelines.
Aesthetic Considerations:
Corrosion can also affect the appearance of a vehicle. For EV manufacturers who emphasize design and aesthetics, ensuring that the vehicle maintains its visual appeal over time is crucial for customer satisfaction.
To mitigate and address corrosion issues in EVs, manufacturers take the various measures, including the use of corrosion-resistant materials, applying protective coatings and sealants to prevent moisture and salt exposure, regular maintenance and warranty coverage. It’s important for EV owners to follow manufacturer recommendations for maintenance and care, as well as be aware of their local environmental conditions. Proper care and maintenance can help reduce the risk of corrosion and ensure the longevity and safety of the vehicle. Additionally, addressing corrosion issues promptly can prevent them from becoming more serious and costly problems in the long run.
In summary, implementing corrosion protection measures for electronic vehicles should be based on specific circumstances and environmental conditions. Corrosion protection is fundamental for the long-term performance, safety, and sustainability of electric vehicles. It addresses not only technical and safety considerations but also contributes to the overall economic and environmental viability of EVs in the automotive market.
SuperCORR A Specialist Barrier Film Corrosion Protection

EnviroTech Europe supplies advanced corrosion protection products, based on approved synthetic materials, to provide quality solutions to a diverse range of lubrication and corrosion problems. SuperCORR A is a unique and proprietary formulation with long-lasting, anti-corrosion inhibitors providing a superior lubrication coefficient and protection against moisture, wear, general and fretting corrosion, static electricity, corona, and other electro migration problems. The non-flammable film is only 7 microns (0.007mm) in thickness, is not a wax or oil-based product and is formulated without sulphates, chlorides, petroleum-based material, or halogens, to meet the EU RoHS directive.
SuperCORR A is unexcelled in preventing failures of electrical systems and electronic equipment caused by corrosion as well as preventing the corrosion of metal components surfaces, including those of components comprised of dissimilar metals such as those found in aerospace environments.The ability to displace water from exposed contacts can ensure reliable operation in extreme conditions. The use of SuperCORR A for corrosion control can not only bring financial savings in reduced maintenance and replacement costs but more importantly greater safety. It is much simpler and a lot less costly to prevent corrosion than to repair or replace damaged avionic equipment or a component that failed because of corrosion.
SuperCORR A is packaged in aerosol cans making access to component parts easy for engineering crews in difficult locations and conditions. Unpainted mild steel will not rust on exterior surfaces directly exposed to sea water environments for at least 6 months, protecting electrical connectors, switches, chains, drive shafts from corrosion while maintaining lubrication on moving surfaces.
EFFICIENT AND ECONOMICAL
- Extremely long-lasting, specially formulated and proprietary anti-corrosive inhibitor.
- Eliminates premature failure of components created by moisture, general or fretting corrosion.
- Prevents deterioration and contamination on all surfaces of electronic and electrical equipment and mechanical close tolerance moving components.
- Reliability increased, maintenance intervals increased and costs reduced, manufacturers save costly warranty service calls or product re-call.
HISTORY
SuperCORR A was originally developed for the U.S. Air Force to comply with military specifications and to prevent electrical and electronic components from systems failures caused by corrosion.
It became the industry standard for avionic corrosion protection within MROs (maintenance, repair and operations) and OEMs (overhaul and original equipment manufacturers). It’s unique ability to displace water and provide a performance enhancing level of corrosion protection has led to it being used in many other applications and industries worldwide.
FURTHER INFORMATION
Please visit our website https://www.envirotech-europe.com/supercorr-a for for information about other uses and applications for Super CORR A.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
For more advice, please telephone us on +44 (0) 20 8281 6370 or use our website contact form. All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
A guide to critical cleaning of oxygen components and services

Guide to critical cleaning of oxygen components
The COVID pandemic of 2020-2023 increased the demand for critical cleaning of oxygen components to treat patients with symptoms. The need for respirators and installation of new and extended oxygen systems in hospitals, clinics and care homes with the provision of ancillary pressure reduction and delivery systems led to a massive increase in demand for the valves, fittings and pipelines which control the supplies from storage tanks and cylinders, all of which needed critical cleaning.
CLEANING OXYGEN COMPONENTS
Critical cleaning of oxygen components is a crucial process to ensure the safety and reliability of systems that handle oxygen, especially in industries like aerospace, healthcare, and industrial gas production.
Cleaning for oxygen service is best defined as the removal of combustible contaminants from the surface of any equipment or system in oxygen service. Essentially, any component that may encounter an oxygen rich environment. The combustible contaminants include organic and inorganic substances such as hydrocarbon material for example oils and greases, paper, fibre, dust, and soils. If these contaminants are not removed properly, in a worst-case scenario, this can cause combustion or explosion in an oxygen atmosphere or at the least rejection of the product due to unacceptable product purity.
Oxygen is not flammable, but it supports combustion. Oxygen can react with most materials. The higher the oxygen content and/or pressure in a system the more vigorous the combustion and the lower the ignition temperature required. Materials that do not normally ignite in air will burn and may explode in an oxygen rich environment. In addition, the oxygen rich environment will give rise to a higher flame temperatures and combustion velocity with potentially devastating consequences. The recognition of oxygen’s reactivity has led to stringent requirements regarding the cleanliness of equipment in oxygen service. Strict guidelines exist to ensure that care must be taken in the selection of equipment including all materials and components, which all need to be oxygen compatible. They must also be free from combustible contaminants as described above.
Consideration must be given to any cleaning processes employed in the manufacture and maintenance of all components of oxygen service systems. There are many options depending on the type of contaminants from aqueous to semi aqueous and blasting systems, to removal of welding slag etc. These are all referenced in the section below.
INDUSTRY STANDARDS FOR OXYGEN SERVICE CLEANING
As part of the programme of worldwide harmonisation of industry standards, the ‘European Industrial Gases Association’, (EIGA) published Cleaning of equipment for Oxygen Service Doc 33/18. The publication was developed from the Compressed Gas Association Document G-4.1 Cleaning Equipment for Oxygen Service by the ‘Compressed Gas Association’.
Specific consideration must be given to the following:
- Cleaning standard to be achieved (how clean is clean?).
- Cleaning procedure.
- Solvent cleaner to be used.
- Surface properties of the parts to be cleaned.
- Shape and geometry of the material.
- Types and amounts of contaminants.
- The degree of automation required.
The size and capacity of the equipment is determined by:
- The size of the material or components to be cleaned.
- The required throughput.
Your starting point should be the cleaning standard and procedure. Solvent cleaning and solvent vapour phase cleaning of components consists of the removal of contaminants by immersion in the solvent, possibly with the addition of ultrasonic agitation and the action of continued condensation of solvent vapour on the component surfaces. The procedure requires that the oxygen equipment, system or component is colder than the solvent boiling point. This allows the vapour to condense on the components and perform a final rinse.
The major significant advantage of solvent cleaning is that vaporised solvent is always pure, and the contaminants remain in the boiling liquid section which requires only periodic cleaning out, thus causing a reduction in the frequency of system downtime. The effectiveness of a particular cleaning agent depends upon the method used, the nature and type of the contaminants and the characteristics of the article being cleaned, such as size, shape, and material. Final evaluation of the cleaning agent should include testing of actual products and production processes. All equipment must, together with the cleaning chemistry, comply as a minimum with current legislation for health, safety and environment. The efficiency is controlled by utilising typical samples, written procedures and requested criteria for cleanliness.
THE SOLVENT OF CHOICE FOR THE CRITICAL CLEANING OF OXYGEN SERVICE COMPONENTS
Zero Ozone Depletion Potential (ODP) and very low Global Warming Potential (GWP) solvent cleaner for high performance critical cleaning of oxygen components and services.
ProSolv® 5408e has a unique range of characteristics make it the solvent of choice for critical cleaning applications such as oxygen components and service line cleaning, aircraft avionics and aerospace parts and servicing, precision optics, medical and high vacuum components and electronics.
ProSolv® 5408e is a high-performance solvent cleaner used for sustainable and future proof degreasing. It has a GWP of less than 1, with a 100-year Integrated Time Horizon (ITH). Soft on the environment and safe for users, it offers improved cleaning at lower costs.
ProSolv® 5408e ticks all the boxes and is the perfect profile for a modern degreasing solvent. Exceptionally low surface tension to penetrate micron sized holes and close contact surfaces. Sustainable and secure for the future. Non-carcinogenic, low boiling point, economical with energy with low solvent losses, faster production, reduced costs, easy handling. Exceptionally low surface tension to penetrate micron sized holes and close contact surfaces.
ProSolv® 5408e has a unique range of characteristics making it the solvent of choice for cleaning oxygen components and service line cleaning, aircraft avionics and aerospace parts and servicing, precision optics, medical and high vacuum components and electronics.
ProSolv® 5408e can be used in most solvent cleaning systems, one tank vapour degreasing or multi tank immersion / vapour tank systems using ultrasonics or flush cleaning for complex mechanical and electronic components with blind holes and channels and close mounted electronics or for removing grinding and buffing soils on surfaces.

EFFICIENT AND ECONOMICAL
- Fast precision cleaning with short cycle times.
- Can be used in any vapour degreasing equipment, lower energy consumption and lower maintenance.
- Can be used as a line flushing solvent.
- Ideal replacement for Trichloroethylene, n-Propyl Bromide, Perchloroethylene, high ODP/GWP fluorocarbons and HCFC based solvents.
- Excellent choice to replace flammable solvents such as MEK, Acetone, Isopropyl Alcohol (IPA) or Hydrocarbons.
- Stable with no additives, no testing required.
- Improved productivity, parts exit the machine cool, dry and spot-free with no drying required.
- Fast drying.
- Minimal non-volatile residue (>10ppm).
- High density solution, excellent for ultrasonic cleaning.
- Mid-range boiling point (46°C), processed components easily handled.
- Very low surface tension for penetration into the micron level crevices and holes, efficient cleaning in tight to reach places and complex geometries.
- Easy process monitoring, minimal effort and minimal waste generation, easy reclamation for reuse.
- High Solvency (KB Value 98) for a variety of contaminants.
- Compatible with a broad range of substrates.
SAFE FOR USERS AND THE ENVIRONMENT
- Safe for the environment, Zero Ozone Depletion Potential (ODP).
- Very low Global Warming Potential (GWP).
- GWP of less than 1, AR4 100yr Integrated Time Horizon (ITH).
- Non-hazardous for transportation.
- Non-flammable (No Flash Point).
- Not classified as a carcinogen.
- RoHS compliant.
Listed above are some of the benefits from using ProSolv® 5408e, a sustainable degreasing solvent for critical cleaning and an economical and efficient replacement solution for cleaning systems using older legacy solvents, which are now either banned or being phased out. EnviroTech Europe have many years of experience and our experts are available to guide you through your solvent cleaner changeover procedures or to advise on equipment. We have extensive partnerships with equipment manufacturers through our distributors worldwide and information and advice on solvents and solvent systems and which need to be considered.
FURTHER INFORMATION
Please visit our website https://www.envirotech-europe.com/prosolv5408e for information about other uses and applications for ProSolv® 5408e.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
For more advice, please telephone us on +44 (0) 20 8281 6370 or use our website contact form. All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Related Posts
Latest News
Eliminate bad smells using unique concentrated water based cleaning system

Aquus® CL-One is the ALL purpose concentrated cleaning system based on an innovative formulation of surfactants which also controls odours and will eliminate bad smells at source.
The unique combination of properties exhibited by Aquus CL-One offers excellent emulsion-breaking capability in addition to its unusual combination of properties that makes it so attractive for a wide range of industrial applications including an unusual application which is discussed below in more detail, utilising these properties to control odours and eliminate bad smells at source.
CONTROL ODOUR BY ELIMINATING UNPLEASANT SMELLS AT SOURCE
Aquus CL-One eliminates odour problems caused by bacteria and other organisms. No fragrance or other masking agents are used, the odour is very effectively eliminated at source. Conventional masking agents use fragrances or enzymes which act slowly and are less effective and more expensive than the Aquus CL-One alternative which uses a totally different approach to these problems.
MANY APPLICATIONS FOR ODOUR CONTROL
Odour control is needed where cleaning is performed in food preparation, processing equipment and containers, sewage treatment plants and in other waste-water facilities.
VOLATILE ORGANIC COMPOUNDS
Aquus CL-One breaks down the organic compounds responsible for the release of the volatile organic compounds (VOCs) producing the unpleasant smells associated with some commercial and industrial processes and systems.
Aquus CL-One eliminates the ability for bacteria to produce the odour producing VOCs by disrupting the integrity of the cell walls. Odour producing microorganisms have cell walls made up of organic materials including lipids (fats). The alkalinity and surface-active ingredients of the Aquus formula breaks down the cell walls of the organism which protect the inner workings. Once compromised the bacteria can no longer absorb nutrients and eliminate waste. This kills the organism which then cannot generate the VOCs which produce the smells.
ADVANTAGES OVER OTHER METHODS
Aquus CL-One has considerable advantages over the toxic, fragrance or enzyme based alternative odour control products which only mask the smells. Enzyme based formulas need regular maintenance to keep the enzymes active and work slowly. Masking agents are usually ineffective and expensive.
Aquus CL-One eliminates completely the odours and smells associated with these organic materials including bacteria and other organisms simply and effectively.
ENGINEERING WASTEWATER TREATMENT SYSTEMS
Aquus CL-One is extremely effective in separating oil, water, and solids in drains, conveyor traps and sumps on automatic honing, turning and grinding machines when using water-based lubricant and cooling liquids.
Emulsions are broken allowing cleaned metal dust, shavings and dirt to precipitate to the bottom, the solution of Aquus CL-One and water retains the active cleaner in the middle layer, oil and grease float to the top. The efficiency of oil/water separators in plant waste treatment systems are significantly increased while the disposal of toxic waste is greatly reduced.
FARMING AND AGRICULTURE
 Farming and agriculture require reliable water-based cleaners for the innumerable cleaning applications needed particularly where animal husbandry is involved. Scrupulous cleaning of surfaces on rails, floors and walls is essential for the health of the animals. The unique bacterial destruction abilities described earlier is very effective in odour control in many farming applications
Farming and agriculture require reliable water-based cleaners for the innumerable cleaning applications needed particularly where animal husbandry is involved. Scrupulous cleaning of surfaces on rails, floors and walls is essential for the health of the animals. The unique bacterial destruction abilities described earlier is very effective in odour control in many farming applications
Aquus CL-One is the concentrated water activated cleaner which is used at different dilutions to meet the requirements of the many applications and challenges met in farming and agriculture. Aquus CL-One which is completely biodegradable within 30/40 days, is not toxic and not considered a hazard which is particularly important in sensitive environmental sites and situations. Safe for use with any application equipment such as high or low-pressure washers, spray washers or dip tanks it can be used with mops, brooms etc by operators without needing personal protective clothing.
The unique combination of oleophilic and hydrophilic properties encourage sedimentation of solids from liquids in wastewater separation tanks and as the cleaner solution can be recycled and reused until biodegradability reduces its effectiveness it is very economical in use. As an example, in normal use a one litre bottle is mixed with 40 litres of water to activate the cleaning solution which in turn can be recycled many times.
HOSPITALS AND HEALTHCARE
 Cleaning of soiled and contaminated surfaces in hospitals, nursing homes and other medical facilities is essential to control bacterial contamination. Aquus CL-One cleaner can be used for all cleaning applications which reduces time and costs by removing the need for multiple cleaning products usually needed to deal with the challenges presented by the wide range of soils, oils, blood, body liquids and other contaminants which need to be removed.
Cleaning of soiled and contaminated surfaces in hospitals, nursing homes and other medical facilities is essential to control bacterial contamination. Aquus CL-One cleaner can be used for all cleaning applications which reduces time and costs by removing the need for multiple cleaning products usually needed to deal with the challenges presented by the wide range of soils, oils, blood, body liquids and other contaminants which need to be removed.
Aquus CL-One is safe for the environment and users, contains no solvents and is biodegradable within 30 days. With only one product for all cleaning applications incorrect selection or dangerous uses can be avoided.
FURTHER INFORMATION
Please visit our website https://www.envirotech-europe.com/aquus-cl-one for information about other uses and applications for Aquus CL-One.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
Share this page:
EnviroTech Europe 2022 Brochure now available

Advanced solutions for vapour degreasing, corrosion protection, metal cleaning and surface treatments.
EnviroTech Europe are specialists in metal, plastic and other substrate cleaning, pre-treatment and protection. We offer a range of approved products tailored to your industry, which are cost effective, energy efficient, safe for operators and the environment.
We are pleased to introduce our range of products in a new 2022 products brochure, viewable on our website and downloadable in PDF format.
Our products are used in the following industries:
Electronics and Electrical
Environmental
General Cleaning
Industrial and Manufacturing
Marine and Maritime
Mechanical Engineering
Medical Device Manufacturing
Military
Motorsport
Power Generation
Railways and Automotive
To view our 2020 Product Brochure please visit www.envirotech-europe.com/brochure.
All products are supplied and supported by EnviroTech Europe Ltd.
Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Vapour degreasing solvent for precision metal cleaning

EnSolv® CC-A Vapour degreasing solvent for precision metal cleaning
The EnSolv® CC-A formulation for precision metal cleaning responds to the need for high performance vapour degreasing solvents for critical cleaning in general engineering, aerospace, medical optical and electrical industries.
EnSolv® CC-A removes cutting oils, lubricants, lapping compounds grinding pastes and water based cutting oils or fluids. This complex synergistic blend utilises environmentally compliant solvents combined with selected surfactants to remove oils and lubricants to the highest standards for critical cleaning applications and releases and suspends inorganic solids and soils such as fingerprints, polishing compounds and grinding paste from finely finished and highly polished surfaces such as stainless steel and plated parts, optical lenses and components for medical appliances.
EnSolv® CC-A is a proprietary blend of inhibitors and stabilisers for the solvents to prevent water staining and corrosion of yellow metals such as copper, brass and bronze which can be a problem with some other solvents.
EnSolv® CC-A is non-flammable, stable and specifically formulated for use in any vapour degreasing equipment with the added advantage of lower solvent usage, greater stability and reduced costs. The formulation is compliant with all current environmental and user safety legislation.
Product Benefits
Designed for vapour degreasing
Removes oil, grease and soils fast
Simple drop-in replacement for n-propyl bromide and trichloroethylene
Low surface tension
Reduced costs
Compatible with all metals
User friendly
Increased production
Faster cleaning cycles
Components easily handled after cleaning
Safe for the environment
Talk to us about whether EnSolv® CC-A is the best choice for your application. Advice, literature, changeover instructions are all available to make the change quickly, easily and at no extra cost of usage.
FURTHER INFORMATION
Please visit our website https://www.envirotech-europe.com/ensolvcc-a for information about other uses and applications.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
We can provide you with a Material Safety Data Sheets, independent laboratory reports, product samples and technical assistance.
For more information or advice please telephone us on +44 (0) 20 8281 6370 or use our contact form.
All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Related Posts
Non chlorinated industrial solvent cleaners
Clarea® industrial solvent cleaners – for safe removal of dirt, oil, grease and other contaminants

Clarea® industrial solvent cleaners are a range of highly refined hydrocarbon based
degreasing solvents with very high cleaning power and low odour, which can be used as an
alternative to other hazardous solvents.
Used throughout industry to remove oils, greases, hydraulic fluids, cutting fluids and a wide
variety of hydrocarbon based protective coatings. The solvent cleaners can be used in dip,
brush or in hand-wipe applications.
Clarea® products leave minimal residues on drying surfaces and are compatible with all
metals and many plastics and composite materials. Contains no halogens such as chlorine or
fluorine.
Clarea® HC40 has a relatively fast evaporation rate with low odour and is suited to hand
wipe and brush cleaning and air dries. Flash Point over 40°C.
Clarea® HC62 is a non-carcinogenic solvent mixture that is safe to use in many applications
and is a not classified as flammable. Flash Point over 60°C.
Clarea® industrial solvent cleaning solvents are one of the ranges of metal cleaning and
surface treatment products from EnviroTech Europe Ltd.
EFFICIENT AND ECONOMICAL
- Versatile and cost-effective solutions for all your industrial cleaning
- Minimal odour and low toxicity for operators
- Multipurpose uses can reduce the inventory of cleaning fluids.
- Flexible uses – evaporation rates suitable for hand wipe, spray, or immersion tank cleaning
- Clarea HC40 for brush and hand wiping, air dries. Flash point 40°C
- Clarea HC62 for spray or immersion cleaning Flash point 62°C
- Contains no chlorine, other halogens or stabilisers
- Compatible with all metals and composites and most plastics
- Excellent pre-cleaner for paint or powder coatings
- Safe, reliable, environmentally friendly cleaning
We can provide you with a Material Safety Data Sheets, independent laboratory reports, product samples and technical assistance.
For more information or advice please telephone us on +44 (0) 20 8281 6370 or use our contact form.
All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Related Posts
SuperCORR A -Advanced corrosion protection for aircraft

Advanced corrosion protection for aircraft – SuperCORR A lubricates and protects mechanical and electrical flight controls in surveillance aircraft.
Super CORR A provides advanced corrosion protection for aircraft – It lubricates and protects mechanical and electrical flight controls and electronic equipment in Lockheed Martin P-3C Orion aircraft flown by air forces around the world for anti-submarine and maritime surveillance, also used in searches for missing ships or aircraft.
The Royal Australian, Korean and Japanese Air Forces were employed in the search for the Malaysia Airlines flight MH370 Boeing 777 passenger jet which disappeared on a scheduled flight in March 2014. The search for debris in a vast area of ocean, bigger than the land area of Europe, southwest of Perth, Australia operated for thousands of hours at low levels but failed to locate any debris and was suspended in 2017.
The maintenance schedules specify Super CORR A to lubricate and protect the flap tracks and screw jacks in the wings of the aircraft which are constantly exposed to extremely corrosive salt laden air as the aircraft in search mode can be flown at very low altitudes of hundreds of feet for long periods.
Super CORR A lubricant and protective coating is widely used by the military, electrical, electronics and aerospace industries for critical applications to give the protection needed in extreme conditions. Super CORR A deposits a dry protective film with excellent corrosion protection and lubricant properties plus the added advantage of a hydrophobic surface rejecting water that ensures the easiest and best connections for very close spaced connectors, contacts, wiring and moving parts
The U.S. Navy selected Super CORR A after exhaustive testing using American Society for Testing and Materials (ASTM) Standard B117- Standard Practice for Operating Salt Spray Apparatus.
Super CORR A, a Type I, Grade B Corrosion Preventative lubricant out-performed 11 other products in comparative testing to identify the best product to protect and lubricate the flap tracks and screw jacks on the aircraft.
The flap tracks, located in the aircraft’s wings, are what the flaps slide on when they move up or down to lower or increase speed. The screw jacks engage and retract the wing flaps. Corrosion on any of the surfaces can lead to snatching or vibration which can affect the pilot’s control
Due to constant operation in salty and corrosive atmospheres which need post-flight rinses and monthly washing down of the aircraft re-lubrication and corrosion treatment for the flap tracks and screw jacks is required each time, with conventional lubricants, to prevent rusting. Super CORR A was evaluated against competitive products under the Federal Test Method Standard #791B, using a five percent Salt Spray Corrosion Test.
Super CORR A far exceeded the performance of the other products evaluated. After over 200 hours of continuous exposure to salt spray corrosion, Super CORR A protection actually increased over time where all the other, similar, products failed early or contributed to an increase in corrosion.
The accelerated salt fog corrosion testing demonstrated that the application of
Super CORR A which complies with MIL-DTL-87177B (revised MIL-L-87177A) also increased electrical operation to 1400 hours versus 100 hours for the control product used at that time. Technical manuals were updated to include the application for electrical and mechanical parts for civilian and military operations.
The most important conclusion from the historical data and prototype testing is the availability of this excellent corrosion preventive compound that has dry film lubricant properties. The application of Super CORR A on aircraft components can reduce maintenance man hours, reduce part replacement costs, increase life of aircraft, safety, and readiness for operations.
Estimates for the maintenance cost for the US Air Force F-16 fleet can reach $500 million per year, the use at all military branches could reach billions of dollars per year. Applications at locations tested by the U.S. Air Force are not normally treated with corrosion prevention and control lubricants (CPC’s). These are the electrical connectors that are susceptible to subtle and not so subtle forms of corrosion that can interfere with the electrical operation of the F-16.
Testing by the U.S. Navy at NADEP Jacksonville incorporates not only electrical connectors, but mechanical and structural components as well. Future uses will also include ground support equipment. The properties of Super CORR A are such that it can be used in a wide variety of applications and on any materials, metal or plastic. Properties of the Super CORR A far exceed the requirements defined by the MIL-L-87177A specification. Many of the beneficial properties of the product are not required in the MIL specification.
EnviroTech Europe is a manufacturer and distributor of anti-corrosion, lubricants and coatings used on wiring, connectors, switches, circuitry, avionics. Read the full report – ‘Use of MIL-L-87177A as a corrosion prevention compound’ in our Library https://www.corrosion-protect.com/resources
Please visit our website https://www.envirotech-europe.com/supercorr-a for for information about other uses and applications for Super CORR A.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
We can provide you with a Material Safety Data Sheets, independent laboratory reports, product samples and technical assistance.
For more information or advice please telephone us on +44 (0) 20 8281 6370 or use our contact form.
All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Aquus CL-One general purpose industrial cleaning agent

Aquus® CL-One concentrated general purpose industrial cleaning agent –
Non-toxic, water based cleaning and protection of metal surfaces – formulated to offer one solution for all cleaning applications in manufacturing, engineering and general maintenance.
Aquus CL-One is a non-toxic general purpose cleaning agent based on a mixture of surfactants and exhibits a unique combination of characteristics not found in other comparable commercially available products.
Aquus CL-One is formulated to offer ONE SOLUTION for all cleaning applications and reduces time and costs by removing the need for multiple cleaning products usually needed to deal with the challenges presented by the wide range of soils, oils and other contaminants which need to be removed.
Aquus CL-One is biodegradable, non-flammable, non-toxic at used concentrations and, unlike almost all other surfactant systems, is both hydrophilic (water attracting) and oleophobic (oil-repelling).
Aquus CL-One offers excellent emulsion-breaking capability in addition to its unusual combination of properties that makes it so practical for a wide range of industrial applications.
Safe for the environment and users, contains no solvents and is biodegradable within 30 days. One of the innumerable industry specific applications are discussed below but many more will be experienced in day-to-day operations. With only one product for all cleaning applications incorrect selection or dangerous uses can be avoided.
Concentrated Aquus CL-One chemistry is activated by water, varying concentration levels allows the most economical solution to be chosen for specific applications from parts cleaning to surface preparations for finishing or heavier soiling on process machines all from one product. Some of the applications are discussed below.
MANUFACTURING, ENGINEERING and MAINTENANCE
Formulated to tackle the numerous cleaning applications required by the manufacturing and engineering industries. Concentrated Aquus chemistry is activated by water, varying concentration levels allows the most economical solution to be chosen for specific applications from parts cleaning to surface preparations for finishing or heavier soiling on process machines all from one product. Some of the applications are discussed below. Our applications experts and samples of the product are available by completing the contact form.
PARTS AND COMPONENT CLEANING
Ideally suited for cleaning protective coatings, grease and soils from used or parts during manufacturing processes. Aquus CL-One can be used in parts washing equipment or dipping tanks without modifications and leaves no residue after drying. Oil and grease separate from the water and coalesces on the surface of the cleaning solution which can then be skimmed off for recovery and recycling. Precipitated solids free of hydrocarbons can be removed by settlement or in filtration systems. The cleaning solution can be reused continuously within its chemical effectiveness parameters for efficient and economical use.
GENERAL CLEANING
Aquus CL-One is remarkably efficient removing oils, greases, and accompanying dirt from most surfaces. It can be applied by wiping surfaces, mopping floors or in rotary floor cleaning machines, or by low or high-pressure spray. Aquus CL-One is also an effective cleaner in industrial kitchens removing grease and baked on deposits on internal surfaces of ovens and trays. For maintenance of machines, engines etc oil and grease is easily removed pre and post assembly making inspection and repair easier, quicker and cheaper by returning equipment to use.
WASTE TREATMENT SYSTEMS
Aquus CL-One is extremely effective in separating oil, water, and solids in drains and conveyor traps and sumps on automatic honing, turning and grinding machines when using water based lubricant and cooling liquids. The emulsions are broken allowing cleaned metal dust, shavings and dirt to precipitate to the bottom, the solution of Aquus CL-One and water retains the active cleaner in the middle layer, and oil and grease float to the top. The efficiency of oil/water separators in plant waste treatment systems are significantly increased while the amount of toxic waste requiring disposal after treatment is significantly reduced. The cleaning solution can be reused to treat more waste streams until biodegradation occurs and it no longer operates effectively.
FURTHER INFORMATION
Please visit our website https://www.envirotech-europe.com/aquus-cl-one for information about other uses and applications for Aquus CL-One.
Visit www.envirotech-europe.com/applications-and-case-studies for information about uses and applications for all EnviroTech Europe products.
We can provide you with a Material Safety Data Sheets, independent laboratory reports, product samples and technical assistance.
For more information or advice please telephone us on +44 (0) 20 8281 6370 or use our contact form.
All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Aquus CL-One Concentrated all-purpose cleaner

Aquus® CL-One concentrated all-purpose cleaner – Water based cleaning and protection of metal surfaces prior to powder coating or painting.
Aquus CL-One is an excellent all-purpose cleaning system for the cleaning and protection of mild steel and ferrous metal parts which need powder coating or painting immediately or when it is necessary to store unpainted for limited periods. Any cleaning system can be used – hand wiping, dip tanks, ultrasonics or pressure washing.
Uncleaned metal components or fabrications before cleaning are protected by process oils and grease on the metal guarding the surfaces from changes in humidity. Once removed using conventional water-based cleaners flash rusting can occur very quickly during drying.
The unique Aquus CL-One formulation however leaves an invisible barrier on the metal surfaces, after the cleaning process, providing extended corrosion protection during storage, fabrication, or assembly before finishing. The barrier film can be powder coated or painted over without additional rinsing and without affecting adhesion of the coatings.
General purpose cleaning
Aquus CL-One is also an excellent general-purpose concentrated surfactant system containing no solvents which exhibits a unique combination of characteristics that are not found in other commercially available cleaning processes biodegradable, non-flammable, non-toxic at used concentrations and, unlike almost all other surfactant systems, is both hydrophilic (water attracting) and oleophobic (oil-repelling).
Aquus CL-One offers exceptional emulsion-breaking capability in addition to its unusual combination of properties that makes it so attractive for a wide range of industrial applications. In addition to the general cleaner Aquus CL-One, other concentrated cleaners within the range are formulated to offer cleaning solutions for industry specific applications.
One cleaner for all applications
One cleaner for all applications reduces costs by removing the need for multiple cleaning products usually needed to deal with a wide range of soils or contaminants. Safe for the environment and users, containing no solvents is biodegradable within 30 days. Typical with only one product for all cleaning applications incorrect selection or dangerous uses can be avoided.
Please visit our website https://www.envirotech-europe.com/aquus-cl-one for information about other uses and applications.
We can provide you with a Material Safety Data Sheets, independent laboratory reports, product samples and technical assistance.
For more information or advice please telephone us on +44 (0) 20 8281 6370 or use our contact form.
All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page:
Corrosion protection for unpainted metal in highly corrosive environments
Tests demonstrate the amazing properties of SuperCORR A corrosion protection spray to protect unpainted metal surfaces from corrosion

These are photographs of 3 unpainted copper components that were exposed for a month next to the “digester” at a Pulp and Paper Mill. The part marked #1 was the control and not sprayed with SuperCORR A corrosion protection spray. Half of part marked #2 was sprayed with SuperCORR A and half left uncoated. Component #3 was completely treated with SuperCORR A. The main atmospheric corrosion was from hydrogen sulphide (H2S) gas, produced during the cellulose digestion process, which combines with the atmospheric humidity to form a mild acid. Any unpainted or untreated metal surface, copper, brass, mild steel or alloys of zinc and manganese or magnesium will be corroded by this acid when it condenses on the metal surfaces.
This is especially important in electrical motors and power control systems where these metals are used as conductors or switch contacts on printed circuit tracks and all types of connectors which although sealed in control panels will be exposed whenever the boxes are opened or contactors operated and connectors plugged and unplugged. In most cases it is not possible to paint these conductive surfaces or apply other forms of protection without loss of conductivity leading to arcing or supply failures.
These tests demonstrate the amazing properties of SuperCORR A to protect unpainted surfaces from acidic corrosion. As an example, copper and aluminium condensers on water coolers usually last about a month in the acidic environment of pulp production facilities because the corrosive atmosphere around the plant corrodes the metal surfaces on fins and connections.
A trial was set up by the works engineering department at the plant who sprayed a new condenser with an aerosol of SuperCORR A corrosion protection spray. This applies a very thin protective film of a proprietary formulation containing extremely long-lasting anticorrosion inhibitors, which also give superior lubrication coefficients and protect components against moisture. This unique, water displacing lubricant, with synthetic corrosion prevention compounds is tested and proven non-flammable and environmentally friendly without sulphates, chlorides, or other halogens
After a few months the treated condenser showed so little signs of corrosion that further controlled trials were set up to test for long term protection of untreated metal in this extremely corrosive environment experienced in paper and pulp mills, water processing plants and oil drilling rigs.



The improvement is clear and demonstrates the savings in costs of parts and labour for maintenance and replacement untreated metal components exposed to these corrosive conditions.
For more information please visit www.envirotech-europe.com/supercorr-a
To discuss the use of SuperCORR A to solve your problems please use the website contact form or phone us, we look forward to hearing from you.
We can provide you with a Material Safety Data Sheets, independent laboratory reports, product samples and technical assistance.
For more information or advice please telephone us on +44 (0) 20 8281 6370 or use our contact form.
All products are supplied and supported by EnviroTech Europe Ltd. Manufactured in the United Kingdom and available on short delivery times through our dedicated team of distributors worldwide.
Share this page: